

  |
|
鄭永祥(1) 龐 飛(2) 摘 要 本研究之目的在於嘗試自周邊血液分離及純化鴨淋巴細胞,探測其對各種淋巴細胞裂殖原刺激之反應,並藉以探討黃麴毒素對鴨淋巴細胞轉型作用之影響。 將EDTA抗凝之成熟產蛋菜鴨血,於4℃下,1300×g,離心30分鐘,取Buffy coat,經等量PBS稀釋後,置於Ficoll-Paque上層,於4℃下,200×g,離心25分鐘後,取界面層可得鴨淋巴細胞再經PBS沖洗一次,RPMI清洗培養液沖洗兩次,每次均於4℃,200g,離心10分鐘。細胞經cytospin後製成抹片,以Quick stain染色計數,所得鴨淋巴細胞純度可達95%以上,平均每10ml血液約可分離2×107細胞。取鴨淋巴細胞(8×105/ml)分別加入最終濃度為0、5、10、20、40或80μg/ml之裂殖原(mitogen),包括有PWM、PHA、PWA及BSS;經培養72小時後,以3H-thymidine (1μCi/well)併入法測定鴨淋巴細胞轉型(lymphocyte transformation)反應。結果以PHA5及10μg/ml濃度下所測得CPM值高於其他裂殖原。 為探討黃麴毒素對鴨淋巴細胞轉型作用之影響,取鴨淋巴細胞(8×105 cells/ml),以PHA (100μg/ml)為陽性對照組,分別加入104、101或10-2ng/ml的黃麴毒素B1;為瞭解微粒體代謝作用對黃麴毒素毒性之影響,於各測試組中加入有或無鴨肝臟微粒體(1mg/ml)與NADPH (0.01mM)二組,經培養後,結果以黃麴毒素101及10-2ng/ml測得之CPM值顯著高於其他各組(P<0.05),顯示低濃度黃麴毒素對鴨淋巴細胞增殖具有刺激作用,輕微的抑制現象僅在高濃度104ng/ml下始出現。反之,當有微粒體存在時,即使在低濃度10-2ng/ml下對鴨淋巴細胞轉型作用即有負面影響,而此抑制作用隨黃麴毒素濃度升高而愈明顯。因此,黃麴毒素在由肝臟微粒體的代謝後,可顯著抑制PHA所引發的鴨淋巴細胞轉型作用。 關鍵詞:黃麴毒素,淋巴細胞轉型作用,鴨。 ───────────────────────────────────── 1.國立宜蘭農工專科學校畜產科講師。 2.國立臺灣大學獸醫學系教授。 (1994年11月22日收件;1995年2月7日接受) 前 言 台灣的高溫多濕,提供了飼料中黴菌生長極有利的條件,飼料中黃麴毒素污染問題一直是相當敏感的事情,雖然高劑量之黃麴毒素污染情形極少,但低量污染卻仍然時有所聞(1)。 黃麴毒素乃黃麴菌中Aspergillus flavus及Aspergillus parasiticus之二次代謝產物。黃麴毒素題黴菌毒素中最重要之一種,為一群結構相似,自然存在之𨧤喃氧雜𤆥鄰酮(furnanocoumarins)(7),曾造成1960年英國爆發10萬隻火雞死亡,稱之為火雞不名疾病 (turkey 'X' disease)(3)。黃麴毒素除了一般人熟知之肝毒性及致癌性之外,尚具有免疫抑制性(20)。報告指出黃麴毒素具有抑制多種非特異性和特異性免疫反應及細胞免疫功能的作用(10),如抑制伴刀豆球蛋白所激發之淋巴細胞轉型作用(2),降低雞網狀內皮系統清除碳粒能力(15),抑制雞血液單核球之趨化性及擾亂固著性及游走性吞噬細胞的吞噬作用(6,19),甚至可進而造成疾病抵抗力的下降(21,24)。 綜觀上述黃麴毒素導致家畜禽免疫能力下降之研究大多集中於豬和雞,但對鴨之研究則闕如。鴨雖具耐粗食和抗病力強等特性,但對黃麴毒素卻甚為敏感(16),本實驗之目的係嘗試分離及純化鴨淋巴細胞,並探討黃麴毒素對鴨淋巴細胞經淋巴球裂殖原(mitogen)所激發的轉型作用(lymphocyte transformation)之影響,以期對黃麴毒素對鴨免疫力之影響有初步的了解。 材料與方法 實驗動物:以成熟的產蛋菜鴨做為血液供應鴨,採血期間鴨隻採籠飼,飼料及飲水均自由採食。 鴨淋巴細胞之分離與純化:採用之方法係依Higgins(9)所發表者予以修改。先自鴨翼下靜脈採血,並以EDTA (0.2%)抗凝。該血液,經4℃,1300×g下離心30分鐘,收集buffy coat,與含0.2% EDTA之等量1×PBS (pH7.2,41℃)混合。再將之置於等量Ficoll-Paque (density:1.077;Sigma, St Louis, Mo., USA)上於4℃,200×g下離心25分鐘,將細胞層吸出,先以1×PBS清洗一次,於4℃,200×g下離心10分鐘後再以含EDTA(0.2%)之RPMI-1640清洗兩次,取得高純度之淋巴細胞。除以trypan blue (0.2%)行vital stain測定細胞存活率外,並取適量細胞液以細胞抹片離心機(Shandon cytospin, Shandon Southern Instruments, Sewelicky, PA, USA)製成抹片,經Diff-Quick染色(International Reagents Corp, Japan)後於顯微鏡下計數200個細胞,測定細胞之分類。依上述結果,將細胞懸浮液以含20%胎牛血清、青黴素(100U/mL)+鏈黴素(100μg/mL)及1%L-glutamine之RPMI-1640調整最終細胞濃縮為1×107淋巴細胞/mL。 裂殖原之製備:本研究中選用PHA (phytohaemagglutinin)、PNA (peanut agglutinin)、PWM (pokeweed mitogen)及BSS (bandeiraea simplicifola seed)等四種裂殖原。先以含20%非動化胎牛血清之培養用RPMI-1640調整為5mg/mL之原備用。 肝臟微粒體之製備:取鴨肝臟40g置於200ml冰冷之1×PBS中,將肝臟剪碎。使用均質機(Ace homogenizer, Nissei AM-5)予以均質,再以組織研磨機(Wheaton overhead stirrer tissue)研磨來回三次。均質液以10000×g離心15分鐘(HIMAC, CR21)使細胞核及粒線體沉澱,上清液以100000×g離心60分鐘,倒去上清液後將沉澱之肝細胞微粒體,以1×PBS稀釋為5mg/mL微粒體蛋白質之原液,微粒體蛋白質含量係依Lowry(13)所發表的方法修定測之,以胎牛血清做為蛋白質標準。 黃麴毒素B1製備:將純品AFB1結晶50mg (Serva, Heidelberg, Germany),先以5mL二甲亞楓(dimethyl sulfoxide, DMSO)溶解,再以1×PBS調整為1mg/mL之原液,以鋁箔紙密封遮光置於-20℃保存備用。 NADPH之製備:NADPH (Sigma, St. Louis, MO., USA),以1×PBS調整為0.25mM溶液,再以鋁箔紙密封遮光置於-80℃保存備用。 試驗一:裂殖原對鴨淋巴細胞的轉型作用 實驗中係使用U型之96孔微量平板(microplate),取80uL,1×107 cells/mL鴨淋巴細胞,加入含40%非動化胎牛血清之RPMI-1640 100μL和最終濃度10倍之裂殖原20μL,使各孔總體積為200μL中含淋巴細胞8×105 cells/mL,20%FBS和裂殖原。將微量平板置入41℃,含5%CO2之濕壓恆溫箱中培養72小時,絡止培養前6小時加入含1μC i3Hthymidine (specific activity, 6.7mCi)之RPMI 20μL,細胞經細胞收集器(PHD Cell Harvester, Cambridge, TechnologyInc., Watertown, WA, USA)收集於玻璃纖維濾紙,濾紙置入含2ml閃爍液(0.5g bezene/toulene)之閃爍瓶中,以βcounter (LS 6000 IC, Beckman Inst. Inc., CA, USA)測定細胞所攝入的3H量,結果以CPM (count per minute)值表示。各處理組詳如表1所示。 |
| Table l. The experimental design of the effect of various mitogens on duck lymphocyte transformation | ||||||
| Treatment | ||||||
| Lym1)+ mitogen 0μg/mL | Lym+ mitogen 5μg/mL | Lym+ mitogen 10μg/mL | Lym+ mitogen 20μg/mL | Lym+ mitogen 40μg/mL | Lym+ mitogen 80μg/mL | |
| RPMI
Lymphocytes (1×107 cells/mL) Mitogen* (200μg/ml) Total |
120μL
80μL
0μL
200μL |
115μL
80μL
5μL
200μL |
110μL
80μL
10μL
200μL |
100μL
80μL
20μL
200μL |
80μL
80μL
40μL
200μL |
40μL
80μL
80μL
200μL |
| *Mitogens included BSS、PHA、PNA、PWM.
1) Lym=Lymphocytes. |
||||||
|
試驗二:黃麴毒素B1對鴨淋巴細胞轉型作用的影響 取1×107 cells/mL之鴨淋巴細胞10μL置於微量平板中,除培養液、肝臟微粒體和對照組外其餘各組均加入25μL濃度為100μg/mL之PHA,並依所需於不同處理組中加入肝臟微粒體50μL及NADPH (0.25mM),和不同濃度之AFB1 (104、101、10-2 ng/mL) 45μL,並以RPMI調整使各孔之總體積為250μL、各處理組詳如表2所示,其後之培養方法如同試驗一。 |
| Table 2. The experimental design of the effects of AFB1 on PHA-induced duck lymphocytes transformation | |||||||||
| Treatment * | |||||||||
| CTRL- | CTRL+ | MIC | A4+MIC | A4 | A1+MIC | A1 | A-2+MIC | A-2 | |
| RPMI
Lymphocytes (1×107/mL) PHA (100μg/mL) AFB1 Microsome (5mg/mL) MADPH (0.25mM) Total |
150μL
100μL
-
- -
-
250μL |
125μL
100μL
25μL
- -
-
250μL |
90μL
100μL
-
- 50μL
10μL
250μL |
20μL
100μL
25μL
45μL 50μL
10μL
250μL |
80μL
100μL
25μL
45μL -
-
250μL |
40μL
100μL
25μL
45μL 50μL
10μL
250μL |
80μL
100μL
25μL
45μL -
-
250μL |
20μL
100μL
25μL
45μL 40μL
10μL
|
80μL
100μL
25μL
45μL -
-
|
| *CTRL-=medium control:CTRL+=PHA control:MIC=microsome alone; A4+MIC=AFB1 104 ng/mL+MICROSOME; A4=AFB1 104ng/mL; A1+MIC=AFB1 101ng/mL+microsome;A1=AFB1 101ng/mL;A-2+MIC=AFB1 10-2 ng/mL+MICROSOME;A-2=AFB1 10-2 ng/ml。 | |||||||||
|
統計分析: 實驗所得之 資料先經一般線性式(general linear modle;GLM)進行變方分析,再以鄧肯氏新多項變域法(Duncans new multiple range test)比較各組間差異顯著性。 結果與討論 鴨血液經Ficoll-Paque梯度處理,純化後,細胞抹片計數可得到純度為95±1.5%之淋巴球,單核球2.0±1.2%,顆粒球1.8±0.3%,紅血球1.0±0.2%和血栓球0.5±0.1%。平均每10mL血液約可分離2×107細胞。紅血球之污染在經低張溶液(0.5%,NH4C1)作用3分鐘,均可完全分解,且對淋巴球之活力無影響。經由此法所分離之菜鴨淋巴細胞純度與Higgins and Chung. (8)使用北京鴨分離之淋巴細胞純度相似(83∼93%)。但本試驗中血液先經離心取得Buffy coat;故在Ficoll-Paque的用量較為節省。由於禽類血液的淋巴細胞在純化上有些困難,包括高濃度顆粒球(4萬個以上/μL);以及淋巴球大小與血栓球和紅血球相似(14),以往的報告也因禽類淋巴細胞純化效率差,且過程複雜,而大多僅用Buffy coat,進行禽類淋巴細胞生物學的研究,也因此間接阻礙了禽類免疫學的研究,較高純度的鴨淋巴細胞取得,首先源自Boyum(5)之方法。將取得之全血與等量PBS混合,可降低血液粘度,改善分離效率。本試驗以Buffy coat與等量PBS混合亦可達到降低粘度之效果並改善鴨周邊血液淋巴細胞與Ficoll-Paque的相容性(compatability)。 有關不同裂殖原對鴨淋巴細胞轉型作用之測試,其結果示於表3。表中顯示BSS對鴨淋巴細胞轉型反應最佳濃度為40μg/mL,PHAl為5μg/mL,PNA為40μg/mL,而PWM為5μg/mL。試驗使用之裂殖原,除PHA及PHA之CPM超過40000以上,其餘BSS及PWM之CPM值均低於10000。此與Higgins.(9)之報告指出最佳反應濃度及CPM值分別為BSS 40μg/mL,23415;PHA 20μg/mL,4754; PNA 20μg/mL,6962;及PWM 40μg/mL,11297,有所差異。歸納其可能原因有:血清來源不同,本試驗使用胎牛血清,而Higgins(9)使用成熟鴨血清;其次可能是血栓球的存在濃度,血栓球一般認為具有抑制淋巴細胞對裂殖原轉型作用的反應(23)。此外,鴨種不同可能對裂殖原的反應亦不同,而此點可能係因特異受器的存在與否及存在的量有關。裂殖原可特異性的位於動物細胞表面糖蛋白及糖脂質內之單糖或寡糖,而淋巴細胞的轉型作用不僅有賴適當的受器存在,另外該等受器分布的密度使其能與裂殖原形成交叉鍵結(cross-linking),亦是決定因子之一。故有些裂殖原雖與受器結合,但如受器密度低時,亦無轉形反應產生(18)。由於菜鴨淋巴細胞胞對T細胞刺激原PHA及B細胞刺激原PNA有較佳的轉型反應,但對於B及T細胞均有刺激作用的BSS及PWM的轉型反應卻不佳。由於PHA及PNA能與細胞糖蛋白及糖脂質之N-acetylgalactosamine結合,而此等受器在鴨淋巴球不僅存在且其分佈的密度極適合PHA及PHA與之形成交叉鏈結,因而導致鴨淋巴細胞明顯的有絲分裂的進行(8,9)。 |
| Table 3. Effects of various mitogens at different concentrations on duck blood lymphocyte transformation | ||||||
| 裂殖源 | Cpm values at various concentraction1) | |||||
| mitogen* | 0 | 5 | 10 | 20 | 40 | 80 |
| BSS
PHA PNA PWM |
828±230
853±218 736±244 763±210 |
3705±828
44565±2627 3916±1257 7260±1527 |
4603±787
39926±4579 6730±1662 6390±1630 |
3444±769
31020±4691 14213±2980 1586±344 |
5197±643
24748±5935 41068±6992 2269±1342 |
3585±1313
2845±972 2586±1011 765±322 |
| 1)
CPM:(count per minute).
*BBS:bandeiraea simplicifola; PHA:phytohaemagglutinin; PNA:peanut agglutinin; PWN:pokeweed mitogen. |
||||||
|
Higgins.(9)指出在低濃度之PHA有較佳之轉型反應可能和受器的異質性(heterogenecity)有關。即當反應的裂殖原濃度改變時與糖脂質或糖蛋白質之親和力或結合力(association)即改變。此外,在反應的細胞群中分化細胞攜帶有不同的接受器,故對裂殖原有不同的反應。 有關AFB1對鴨淋巴細胞轉型作用之影響,其結果示於圖1。圖中顯示AFB1在低濃度時(101及10-2ng/mL)對由PHA所引發的鴨淋巴細胞轉型作用具有刺激的效果,且顯著較PHA陽性處理組為高(P<0.05),反觀當AFB1的濃度提升至104ng/mL時,即開始對PHA所引發的鴨淋巴球轉型作用又有抑制的趨勢,雖然此抑制效果在統計學上並不顯著。然而當各AFB1組中加入肝臟微粒體後,無論是101或是10-2均對PHA所引發的鴨淋巴球轉型作用產生極顯著的抑制作用。由以上的結果顯示肝臟微粒體在AFB1對鴨淋巴球轉型作用抑制性上扮演重要的加乘效應,此點和AFB1對火雞腹腔巨噬細胞的毒性相類似(17)。AFB1如未經肝臟微粒體的活化,即使在高濃度下40μg,對火雞腹腔巨噬細胞的附著力、形態學,及吞噬能力均無顯著的影響;反之,經肝臟微粒體代謝後,AFB1在極低的濃度下(1∼5μg)即可造成火雞腹腔巨噬細胞附著力和吞噬能力的明顯下降,且細胞表面呈球狀突起而核亦呈分解狀(17)。AFB1在氧和NADPH的存在下,可被仰賴cytochrome P450的肝臟微粒體mixed function oxidase (MFO)系統所活化(12),其中一個代謝產物,2,3-oxide可和DNA上N7-guanine結合而產生細胞毒性、致癌性及畸胚性等影響(11)。Bodine et al. (4)亦曾指出受裂殖原PHA致活之牛淋巴球能將AFB1進一步代謝為可和DNA結合的細胞突變物質。且抑制活體外淋巴細胞對各種抗原或細胞裂殖原的刺激反應(21,24)。 |
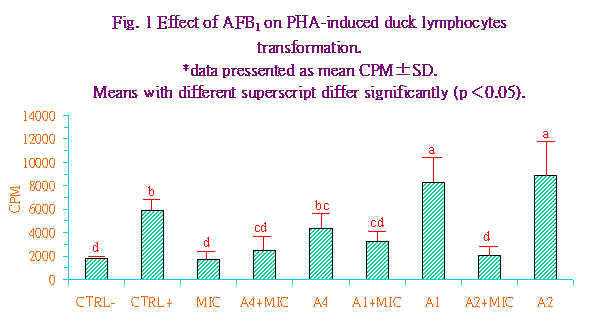
|
綜合本試驗之結果顯示PHA對菜鴨淋巴球有較佳的轉型反應,在無肝臟微粒體存在下低濃度AFB1對該轉型反應具有刺激作用,但AFB1經肝臟微粒體代謝後對轉型反應具有抑制作用。由於鴨較其他動物對AFB1具敏感性;利用鴨隻此一特性探討AFB1對其生理與免疫反應的影響,應是頗佳之動物模式。 誌 謝 本試驗承蒙國立臺灣大學畜產學系陳保基教授提供試驗鴨隻,特此致射。 參考文獻 1.吳繼芳、鄭清森、游義德、嚴家清、郭忠政。1988。飼料中含低濃度黃麴素對生長豬生長性能與組織殘留量之影響。中華農學會報. 144:85-93 2.龐飛。1994。黃麴毒素對豬隻免疫力影響之研究。生命科學簡訊。8(3):1-9. 3.Blount, W. P. 1961. Turkey X disease. J. Brit. Turkey Federation. 9:52. 4.Bodine, A. B., S. F. Fisher and S. Gangjee. 1984. Effect of aflatoxin B1 and major metabolites on phytohemagglutinin stimulated lymphoblastogenesis of bovine lymphocytes. J. Dairy Sci. 67:110-114. 5.Boyum, A. 1968. Isolation of leucocytes from human blood. A two phase system for removal of red cells with methyl cellulose as erythrocyte-aggregatting agent. Scand. J. Clin. Lab. Invest. 21 (suppl. 9),4. 6.Chang, C. F. and P. B. Hamilton. 1979. Impaired phagocytosis by heterophils from chickens during aflatoxicosis. Toxicol. Appl. Pharmacol. 48:459-466. 7.Ciegler, A. and E. B. Lillehoj. 1968. Mycotoxins. Advan. Appl. Microbiol. 10:155-219. 8.Higgins, D. A. and S. H. Chung. 1986. Duck Lymphocytes: I. Purification and preliminary observations on surface markers. J. Immunol. Methods. 86:231-238. 9.Higgins, D. A. 1990. Duck Lymphocytes: III. Transformation responses to some common mitogens. Comp. Immunol. Microbiol. Infect. Dis. 13:12-23. 10.Kadian, S. K., D. P. Monga and M. C. Goel. 1988. Effect of aflatoxin B1 on the delayed type hypersensitivity and phagocytic activity of reticuloendothlial system in chickens. Mycopathologia 104:33-36. 11.Karenlampi, S. O. 1987. Mechanism of cytotoxicity of aflatoxin B1: Role of cytochrome P1-450. Biochem. Biophys. Res. Commun. 145:854-860. 12.Koser, P. L., M. B. Faletto, A. E. Maccubbin, and H. L. Gurtto. 1988. The genetics of aflatoxin B1 metabolism, Association of the induction of aflatoxin B1-4-hydroxylase with the transcriptional activation of cytochrome P3-450 gene. J. Biol. Chem. 263:12584-12595. 13.Lowry, O. H., N. J. Rosebrough., A. L. Far and R. J. Randall. 1951. Protein measurement with folin phenol reagent. J. Biol. Chem. 193:265-275. 14.Lucas, A. M. and C. Jamroz, 1961. Atlas of Avian Hematology, Agriculture Monograph 25 (U.S. Department of Agriculture, Washington, DC). 15.Michael, G. Y., P. Thaxton and P. B. Hamilton. 1973. Impairment of the reticuloendothelial system of chickens during aflatoxicosis. Poult. Sci. 52:1206-1207. 16.Muller, R. D., C. W. Carlson, G. Semeniuk and G. S. Harshfiled. 1970. The response of chicks, ducklings, goslings, pheasants and poults to graded levels of aflatoxins. Poult. Sci. 49:1346-1350. 17.Neldon-Ortiz, D. L. and M. A. Qureshi. 1991. Direct and microsomal activated aflatoxin B1 exposure and its effects on turkey peritoneal macorphage functions in vitro. Toxicol. Appl. Pharmacol. 109:432-442. 18.Pearson, T. W., G. E. Roelants, L. B. Lundin and K. S. Mayor-Withey. 1979. The bovine lymphoid system:binding and stimulation of peripheral blood lymphocytes by lectins. J. Immunol. Methods 26:271-275. 19.Richard, J. L. and J. R. Thurston. 1975. Effect of aflatoxin on phagocytosis of Aspergillus fumigatus spores by rabbit alveolar macrophages. Appl. Microbiol 30:40-47. 20.Pier, A. C., S. J. Cysewski. J. L. Richard, and J. R. Thurston. 1977a. Mycotoxins in human and animal health. Edited by J. V. Rodricks, C. W. Hesseltine, and M. A. Mehlman. Park Forest South, Illinois, Pathotox Publ. pp. 745-750. 21.Singh, J., R. P. Tiwari, G. Singh, S. Singh and D. V. Vadehra. 1987. Biochemical and immunological effects of aflatoxins in rabbits. Toxicol. Letters 35:225-230. 22.Thurston, J. R. M., B. L. Royoe, A. L. Baetz, J. L. Rchard and G. D. Booth. 1974. Effect of aflatoxin of serum protein, complement activity, and the antiboby response to Brucellla abortus in guinea pigs. Am. J. Vet. Res. 35: 1097-1100. 23.Traill, K. N. G. Bock, R. Boyd, G. Wick. 1983. Chicken thrombocytes. Isolation, serological and functional characterisation using the fluorescense activated cell sorter. Dev. Comp. Immunol. 7:112-115. 24.Yang, W. C., 1983. Effects of aflatoxin B1 on the development of procine cellar and humoral immune responses. Ph. D. dissertation, Auburn University, Alabama, USA. |
|
Effects of Aflatoxin B1 (AFB1) on Duck Lymphocyte Transformation Yeong-Hsiang Cheng(1) and Victor-Fei Pang(2) Summary The purpose of this experiment was to isolate and purify duck lymphocytes from peripherial blood. Lymphocytes were then used to study transformation response to various mitogens. These results were used to evaluate the effects of aflatoxin on duck lymphocyte transformation. The buffy coat collected from the whole blood was anticoagulated with EDTA andcentrifuged at 1300×g for 30min at 4℃, then resuspended with an equal wolume of PBS. The cell suspension was layered over a Ficoll-Paque gradient and centrifuged at 200×g for 25min at 4℃. The band formed at the interface of the RBC layer contained cells of which greater than 95% were lymphocytes. The averaged numbers of lymphocytes obtained from 10 mL of whole blood wasapproximately 2 ×107. Various mitogens (PWM, PHA, PNA, and BSS) at a final concentration of 0,5,10,20,40and 80μg/mL were added to the lymphocyte suspension (8×105 cells/mL). The Cpm value of the lymphocyte transformation response by 3H-thymidine integration after 72hrs incubation was measured. The results showed that PHA at 5 and 10μg /mL gave higher values than the others. The effects of aflatoxin on duck lymphocyte transformation response were evaluated, by addition of final concentration of 104,101, or 10-2 ng/mL aflatoxin B1 (AFB1) to lymphocyte suspension. Meanwhile, lymphocyte suspension with or without duck liver micorosome plus NADPH was also evaluated. The results showed AFB1 101 and 10-2ng/mL had significantly higher CPM values than the others (P<0.05). This indicated that AFB1 had a stimulative effect on duck lymphocyte transformation. AFB1 only at 104ng/mL had a slight inhibition. When liver microsome was present, AFB1 had an adverse effect on duck lymphocyte transformation even though at a low concentraction of 10-2ng/mL. This detrimental effect became more obvious as AFB1 concentration increased. It could thus be concluded that PHA-induced duck lymphocyte transformation was inhibited significantly when AFB1 was metabolized by duck liver micorsome, Key words:Aflatoxins, Lymphocyte Transformation, Duck ──────────────────────────────────── 1.Department of Animal Science, National I-Lan Institute of Agriculture and Technology, I-Lan Taiwan, R.O.C. 2.Department of Veterinary Medicine, National Taiwan University, Taipei, Taiwan, R.O.C. (Received November 22, 1994; Accepted February 7,1995)
|